China National Standard for Aluminium-Plastic Combination Caps for Injection Glass Vials for Antibiotics (YBB00372003-2015)
This standard applies to non-sterilized aluminum-plastic combination caps for injection glass vials for antibiotics.
Appearance
Take an appropriate quantity of the product and visually inspect it under bright natural light. The product shall be clean and free from residual lubricant, burrs, damage, and injection molding flash. The plastic component shall be firmly and integrally bonded to the aluminum component.
Mechanical Properties of Aluminum Material* Tensile strength: 100–180 N/mm². Elongation at break: Not less than 2.0%
Tensile strength refers to the maximum tensile load borne per unit of original cross-sectional area during a tensile test until fracture.
Elongation at break refers to the ratio of the increase in gauge length at fracture to the original gauge length, expressed as a percentage.
Test Method:
Take an appropriate amount of aluminum sheet material from the same batch.
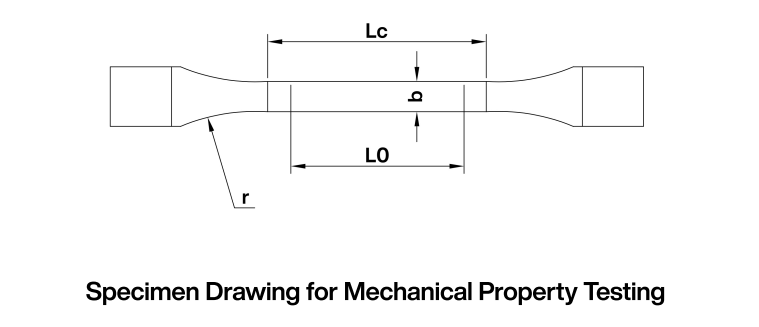
Prepare test specimens using a cutting tool meeting the following specifications (as shown in Figure 1):
Width (b): 12.5 mm
Original gauge length (L₀): 50 mm
Parallel length (Lc): 75 mm
Transition arc radius (r): ≥ 20 mm
Conduct the tensile test on a tensile testing machine at a test speed of 10 mm/min ± 2 mm/min.
The specimens shall be conditioned at 23°C ± 2°C and relative humidity 50% ± 5% for not less than 4 hours prior to testing, and the test shall be performed under the same conditions.
Calculation of Elongation
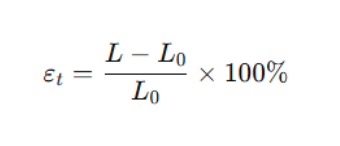
Where:
εₜ = elongation at break (%)
L₀ = original gauge length (mm)
L = gauge length at fracture (mm)
Burr (Edge Protrusion)
Take an appropriate quantity of the product and measure with a vernier caliper accurate to 0.1 mm. The burr shall not exceed 3% (see Figure 2).
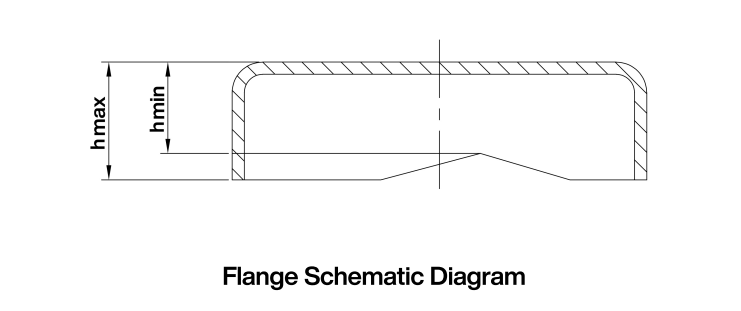
The burr at the edge of the aluminum part shall be expressed as a percentage and calculated as follows:
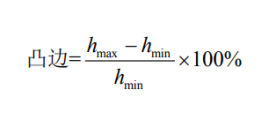
Where:
hₘₐₓ = maximum outer height of the aluminum part (mm)
hₘᵢₙ = minimum outer height of the aluminum part (mm)
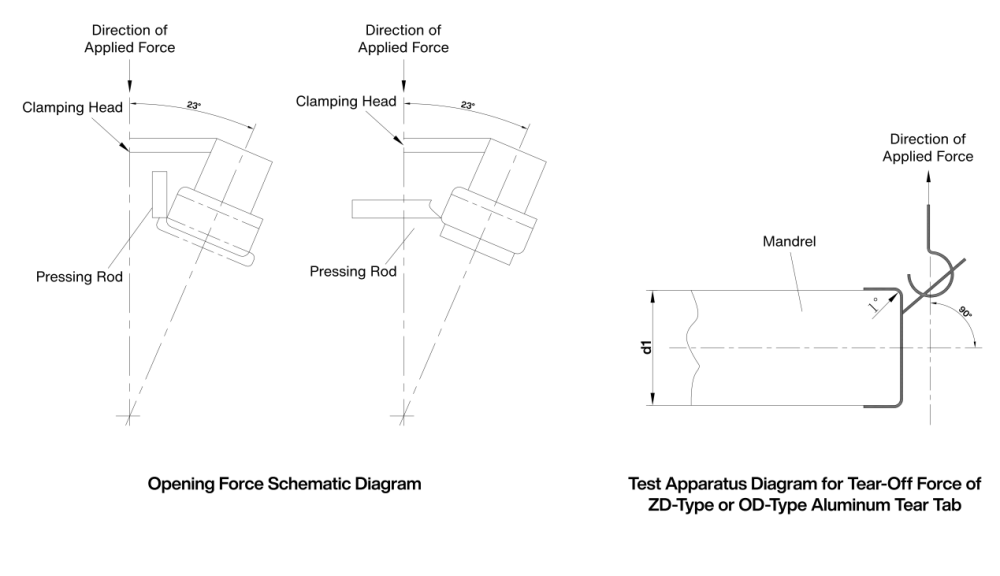
Opening Force
Plastic Component Removal Force
Take an appropriate quantity of the product and test it on a tensile testing device (see Figure 3) at a test speed of 100 mm/min ± 10 mm/min. It shall comply with the requirements specified in Table 1.
Tear-Off Force (ZD or OD Type Aluminum Tear Tab)
Take an appropriate quantity of caps. Hook the pulling device onto the exposed tear tab and perform the test on a tensile testing device (see Figure 4) at a test speed of 100 mm/min ± 10 mm/min until the cap is completely torn off.It shall comply with the requirements specified in Table 1.
|
Table 1 Opening Force |
|||
|
Specification (mm) |
Minimum Plastic Component Removal Force(N) |
Maximum Plastic Component Removal Force(N) |
Maximum Tear-Off Force (N) |
|
13 |
6 |
25 |
30 |
|
20 |
6 |
35 |
40 |
Opening Quality
After performing the opening force test and removing the plastic component, visually inspect the opening of the aluminum part.The opening shall not be damaged.
Compatibility
Take an appropriate quantity of the product and cap it onto a suitable vial filled with labeled volume of water (with rubber stopper applied). Seal it using a capping device. The cap shall fit properly.
Sterilization Resistance
Take an appropriate quantity of the product and cap it onto a suitable vial filled with labeled volume of water (with rubber stopper applied). Seal using a capping device. After sealing, place the vial in a steam sterilizer and sterilize under the following conditions:
121°C ± 2°C for 30 minutes
Including 130°C ± 2°C for 5 minutes
After sterilization, the plastic component shall withstand steam sterilization at 130°C without deformation or discoloration. The aluminum surface shall show no obvious change. The cap shall not exhibit cracking or abnormal deformation.
Coating Adhesion
Take an appropriate quantity of coated products (external surface coated). After steam sterilization at:
121°C ± 2°C for 30 minutes
Including 130°C ± 2°C for 5 minutes
Remove the plastic component. Wipe the surface for 30 seconds using absorbent cotton soaked in 80% ethanol solution, then wipe for 30 seconds using absorbent cotton soaked in 70% isopropanol solution. The coating shall show no wear or abrasion.

Appendix I – Inspection Rules
1. Product inspection includes:
-Full inspection
-Partial inspection
2. Full inspection shall be conducted under any of the following circumstances:
-Product registration
-Resumption of production after a major quality accident
-Supervisory sampling inspection
-Resumption of production after production suspension
3. After product registration approval, if there are no changes in raw material origin, additives, production process, etc., pharmaceutical packaging material manufacturers and users may conduct inspection of all items except those marked with “*”, according to the standard.
4. Appearance, burr, opening force, opening quality, compatibility, sterilization resistance, and coating adhesion shall be inspected in accordance withGB/T 2828.1-2012.
Sampling Procedures for Inspection by Attributes – Part 1: Sampling Schemes Indexed by Acceptance Quality Limit (AQL) for Lot-by-Lot Inspection.
Inspection items, inspection levels, and AQL values shall comply with Table 2.
Note: Items marked with “*” shall be tested at least once every six months.
|
Table 2 – Inspection Items, Inspection Levels, and Acceptance Quality Limits (AQL) |
||
|
Inspection Items |
Inspection Levels |
Acceptance Quality Limits (AQL) |
|
Appearance |
I |
4.0 |
|
Burr / Edge Protrusion |
S-3 |
2.5 |
|
Opening Force |
S-2 |
4.0 |
|
Opening Quality |
S-2 |
4.0 |
|
Compatibility |
S-2 |
4.0 |
|
Sterilization Resistance |
S-2 |
4.0 |
|
Coating Adhesion |
S-2 |
4.0 |
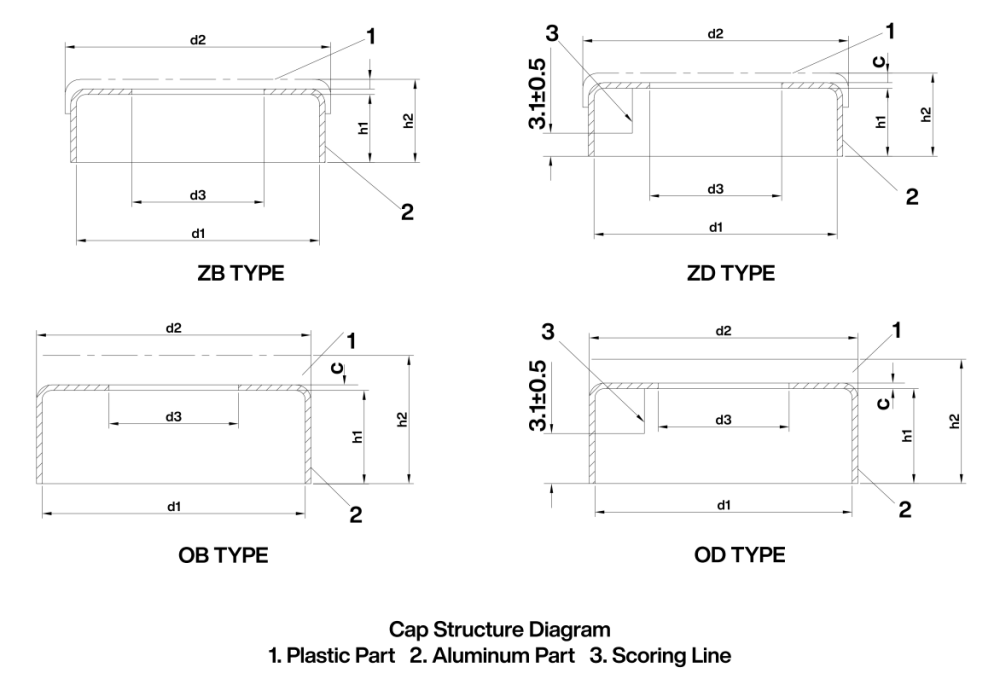
Appendix II – Reference Dimensions
Specification dimensions may refer to Figure 5 and Table 3.
|
Table 3 Reference Dimensions Unit:mm |
|||||||||||
|
Nominal Dimension |
Type |
d1 |
d2 |
d3(Bore Diameter after Plastic Component Removal) |
e |
h1 |
h2 |
||||
|
min |
max |
min |
max |
min |
max |
min |
max |
||||
|
13 |
ZB、ZD |
13.3 |
15 |
16 |
3 |
8 |
0.168 |
0.242 |
6.3 |
7.3 |
8.4 |
|
OB、OD |
13 |
13.8 |
|||||||||
|
20 b |
ZB、ZD |
20 |
22 |
23 |
6 |
10 |
7.3/6.9 |
8.7 |
9.8 |
||
|
OB、OD |
19.7 |
20.6 |
|||||||||
|
20 a |
ZB、ZD |
20.3 |
22.2 |
23.2 |
6 |
10 |
0.168 |
0.242 |
7.3 |
8.7 |
9.8 |
| OB、OD | 20.2 | 20.9 | |||||||||
Notes:
d₂ shall be determined through agreement between supplier and purchaser. Tolerance shall not exceed ±0.25 mm of the nominal value.
e shall be determined through agreement between supplier and purchaser. Tolerance shall not exceed ±0.022 mm of the nominal value.
h₂ shall be determined through agreement between supplier and purchaser. Tolerance shall not exceed ±0.4 mm of the nominal value.
20a cap: Suitable for Type A molded vials and Type A stoppers.
20b cap:
h₁ = 7.3 mm: suitable for Type B molded vials and Type B1 stoppers
h₁ = 6.9 mm: suitable for tubular vials and Type A stoppers
If used with other vial and stopper combinations, the nominal dimension of h₁ shall be determined by agreement between supplier and purchaser within the range of 6.4–7.8 mm.


