China National Standard for Injection Vials Made of Low Borosilicate Glass Tubing (YBB00302002-2015)
This standard applies to low borosilicate glass tubular injection vials intended for direct filling of injectable solutions, sterile powders for injection, and concentrated solutions for injection.
Appearance
Take an appropriate quantity of the product and visually inspect it under bright natural light from a frontal view. The vials shall be colorless and transparent or translucent amber. The surface shall be smooth and even, free from obvious glass defects. Cracks are not permitted in any part of the vial.
Identification*
(1) Coefficient of Linear Thermal Expansion
Take an appropriate quantity of the product and determine the coefficient in accordance with the Average Coefficient of Linear Thermal Expansion Method (YBB00202003-2015) or the Linear Thermal Expansion Coefficient Method (YBB00212003-2015). The result shall be within (6.2–7.5) × 10⁻⁶ K⁻¹ (20–300 °C).
(2) Boron Trioxide (B₂O₃) Content
Take an appropriate quantity of the product and determine the boron trioxide content in accordance with the Boron Trioxide Determination Method (YBB00232003-2015). The content of boron trioxide shall not be less than 5%.
Hydrolytic Resistance of Glass Grains at 121 °C
Take an appropriate quantity of the product and test it in accordance with the Test and Classification Method for Hydrolytic Resistance of Glass Grains at 121 °C (YBB00252003-2015). It shall comply with Class 1 requirements.
Hydrolytic Resistance of Inner Surface
Take an appropriate quantity of the product and test it in accordance with the Test and Classification Method for Hydrolytic Resistance of Inner Surface at 121 °C (YBB00242003-2015). It shall comply with HC1 or HCB grade.
Internal Stress
Take an appropriate quantity of the product and test it in accordance with the Internal Stress Determination Method (YBB00162003-2015). After annealing, the maximum optical path difference caused by permanent stress shall not exceed 40 nm/mm.

Extractable Amounts of Arsenic, Antimony, Lead, and Cadmium*
Take an appropriate quantity of the product and determine the extractable amounts in accordance with the Determination Method for Extractable Arsenic, Antimony, Lead, and Cadmium (YBB00372004-2015). Per liter of extract solution:
Arsenic (As): not more than 0.2 mg
Antimony (Sb): not more than 0.7 mg
Lead (Pb): not more than 1.0 mg
Cadmium (Cd): not more than 0.25 mg
Vertical Axis Deviation
Take an appropriate quantity of the product and test it in accordance with the Vertical Axis Deviation Determination Method (YBB00192003-2015). The results shall comply with the requirements specified in Table 1.
|
Table 1 Maximum Permissible Vertical Axis Deviation |
||||||||||
|
Capacity(ml) |
2 |
3 |
5 |
7 |
8 |
10 |
15 |
20 |
25 |
30 |
|
Vertical Axis Deviation, amax (mm) |
1.0 |
1.2 |
1.5 |
|||||||
Annex I – Inspection Rules
1.Product inspection is classified into full inspection and partial inspection.
2.Full inspection shall be performed under any of the following conditions:
-Product registration.
-Resumption of production following a major quality incident.
-Regulatory sampling inspection.
-Resumption of production after product discontinuation.
3.After product approval and registration, if the pharmaceutical packaging material manufacturer or user maintains the original raw material source, additives, and production process without any changes, inspection may be carried out according to the standard, excluding items marked with “*”.
4.Inspection of appearance, internal stress, and vertical axis deviation shall be conducted in accordance with “Counting Sampling Inspection Procedures – Part 1: Sampling Plans Indexed by Acceptable Quality Level (AQL)” (GB/T 2828.1-2012).
The inspection items, inspection levels, and AQL values are listed in Table 2.
|
Table 2 Inspection Items, Inspection Levels, and Acceptance Quality Limits (AQL) |
|||
|
Inspection Item |
Inspection Level |
Acceptance Quality Limit (AQL) |
|
|
Appearance |
Cracks |
1 |
0.65 |
|
Others |
4 |
||
|
Internal Stress |
S-1 |
1.5 |
|
|
Vertical Axis Deviation |
S-3 |
2.5 |
|
Annex II Dimensional Specifications (Reference Dimensions)
The dimensional specifications may be referenced from Figure 1, Table 3, and Table 4.
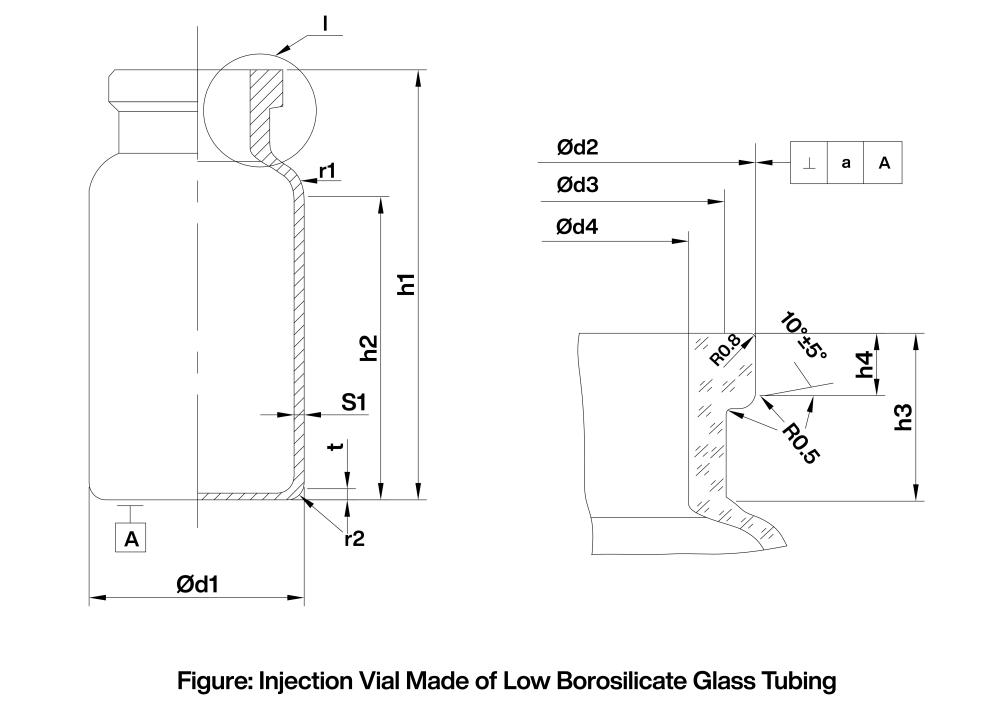
|
Table 3 Main Dimensional Specifications Unit: mm |
||||||||||||
|
Capacity(ml) |
Vertical Axis Deviation, aₘₐₓ |
Bottle Body Outside Diameter, d₁ |
Bottle Mouth Outside Diameter, d₂ |
Bottle Mouth Inside Diameter, d₄ |
Overall Bottle Height, h₁ |
Bottle Mouth Edge Thickness, h₄ |
Bottom Thickness, S₂ |
|||||
|
Dimension |
Tolerance |
Dimension |
Tolerance |
Dimension |
Tolerance |
Dimension |
Tolerance |
Dimension |
Tolerance |
|||
|
2 |
1.0 |
16.0 |
±0.2 |
13.0 |
+0.2 |
7.0 |
±0.2 |
31.0 |
±0.5 |
3.6 |
±0.2 |
≥0.7 |
|
3 |
35.0 |
|||||||||||
|
5 |
18.4 |
7.6 |
39.7 |
|||||||||
| 7 | 1.2 | 22.0 | 19.6 | -0.3 | 12.6 | |||||||
| 8 | 45.0 | |||||||||||
| 10 | 49.7 | |||||||||||
| 15 | 24.0 | 52.0 | ||||||||||
| 20 | 1.5 | 28.0 | ±0.3 | 55.0 | ±0.7 | |||||||
| 25 | 65.0 | |||||||||||
| 30 | 32.0 | 70.0 | ||||||||||
| Table 4 Other Dimensional Specifications (Reference) Unit: mm | |||||||||
|
Capacity |
Maximum Bottle Neck Outside Diameter, d₃max |
Bottle Body Height, h₂ |
Bottle Neck Height, h₃ (Approx.) |
Bottle Wall Thickness, S₁ (Approx.) |
r1≈ |
r2≈ |
tmax |
Mass (Approx., g) |
Brimful Capacity (mL) |
|
2 |
10.5 |
18.0 |
8.0 |
1.0 |
2.5 |
1.5 |
0.7 |
4.2 |
3.5 |
|
3 |
20.0 |
4.6 |
4.2 |
||||||
|
5 |
28.0 |
7.5 |
0.8 |
5.0 |
7.5 |
||||
|
7 |
16.0 |
27.0 |
1.0 |
3.5 |
2 |
8.2 |
10.5 |
||
|
8 |
32.0 |
9.0 |
11.5 |
||||||
|
10 |
37.0 |
9.7 |
13.5 |
||||||
|
15 |
37.0 |
8.5 |
12.1 |
18.5 |
|||||
|
20 |
35.0 |
5.5 |
2.5 |
1 |
15.0 |
23.5 |
|||
|
25 |
47.0 |
16.2 |
28.5 |
||||||
|
30 |
55.0 |
10.0 |
22.5 |
37 |
|||||


